
| Homepage | |
| Group Members | |
| Publications | |
| 2002 - | |
| 1988 - 2001 | |
| Theses and | |
| Reports | |
| Picture Gallery | |
| Data | |
| Databases | |
| Software | |
| Job Vacancies | |
| Conferences, | |
| Workshops | |
| and Courses | |
| Local Info | |
| Group Info | |
| Projects | |
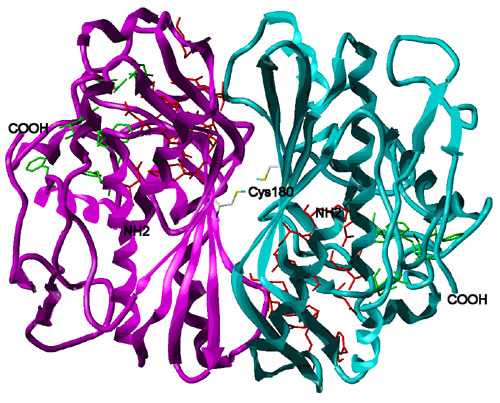 |
To tackle the cellular drug resistance of thymidylate synthase (TS), the protein-protein interactions involved in TS dimerization and mRNA binding are studied, e.g. by using molecular dynamics simulations.
Figure: Dimeric structure of human thymidylate synthase (PDB code 1hzw: [1]). One monomer is coloured in magenta and the other in cyan. Folate-binding site residues are displayed in green and dUMP-binding site residues in red. NH2 denotes the amino terminus and COOH the carboxy terminus of the protein subunits. A cysteine residue that is critical for mRNA binding (Cys180) is located at the dimer interface [2, 3]. 1. Almog R, Waddling CA, Maley F, Maley GF, Van Roey P. Protein Sci. 10: 988-996, 2001 2. Voeller DM, Zajac-Kaye M, Fisher RJ, Allegra CJ. Biophys. Res. Commun. 297: 24-31, 2002 3. Lin X, Liu J, Maley F, Chu E. Nucleic Acid Res. 31: 4882-4887, 2003 |
|
|